Public Sector / Pharmaceutical Regulation
Project Summary
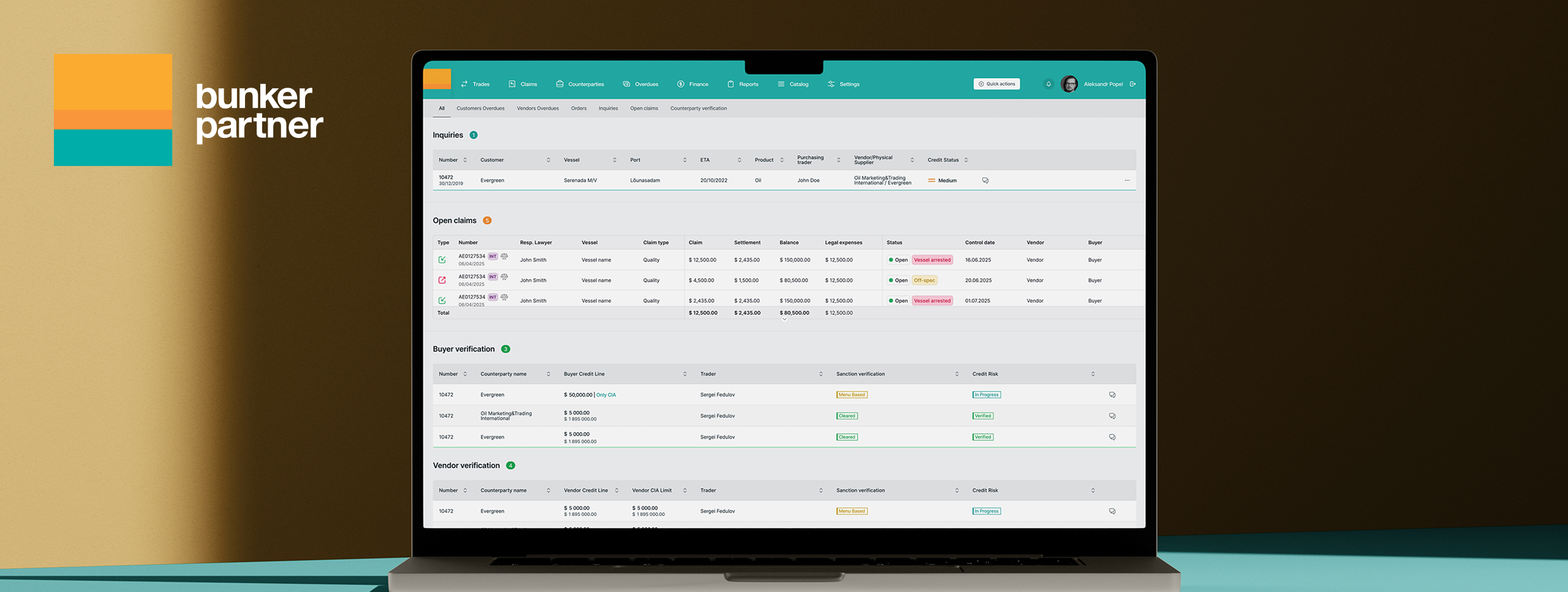
Our team developed and maintains the Estonian Medicines Agency’s digital systems and registers. The platform automates pharmaceutical workflows, patient and provider registries, and public access interfaces — delivering streamlined operations compliant with EU pharmaceutical directives.
Quick Facts & Challenge
Pharmaceutical regulation requires precise data management across medicine registrations, inspections, and compliance reporting. Before modernization, processes were manual and fragmented, limiting efficiency and introducing regulatory compliance risks.
Location
Estonia
Industry
Public Healthcare Regulation / Pharmaceuticals
Period of Development
2018 – 2025
Services Used
System Development and Maintenance, Integration, UX Improvement
Technologies
.NET (C#), Angular (TypeScript), SQL Server, X-Road, eID Authentication, Jenkins
Solution
Integration with X-Road and EU regulatory systems
Public search interfaces for medicine information
Full compliance with EU pharmaceutical directives
The solution’s architecture supports continuous regulatory updates while maintaining system stability and public accessibility.
Results
Streamlined digital workflows for regulatory processes
Improved public access to pharmaceutical data
Seamless EU regulatory compliance
Reduced manual processing and data entry errors
Our team delivered a reliable regulatory platform supporting Estonia’s pharmaceutical oversight.
Client Feedback
Our clients trust us to deliver exceptional results.
“The modernized systems have transformed our agency’s efficiency. Public access to medicine data has never been easier, and our compliance with EU directives is now fully automated.”
Head of IT, Estonian Medicines Agency